Quantum numbers1/5/2024 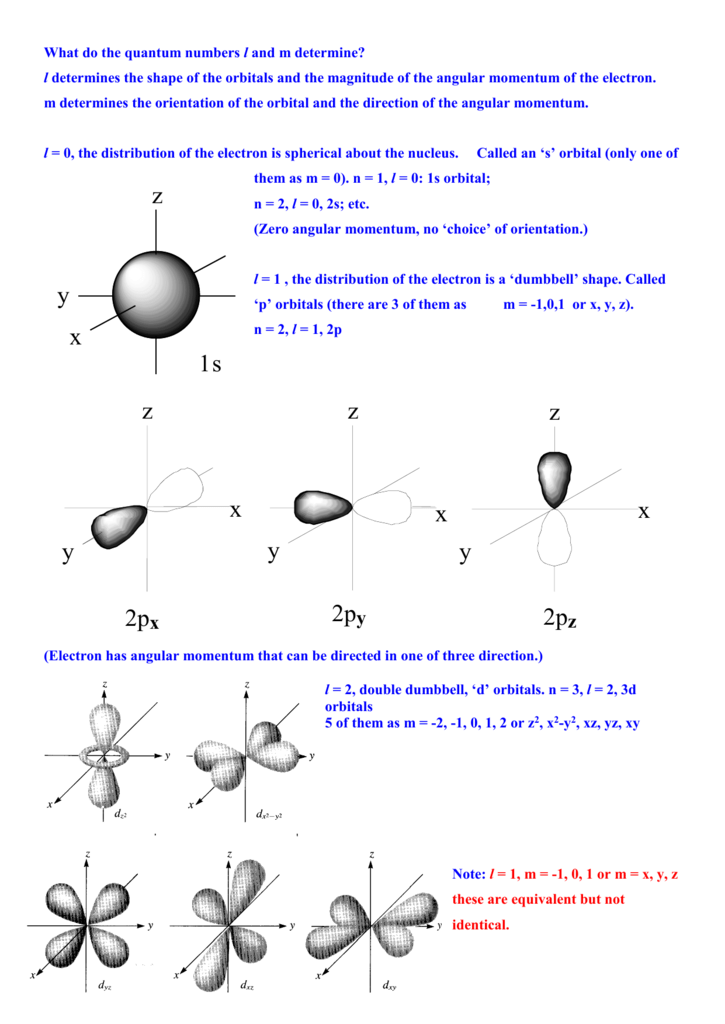 
The shell with principal quantum number of ‘n’, has n 2 orbitals in that shell (energy level). For given value of l, there are 2l + 1 possible values of m l. Where, the subscript represents the direction of axis of rotation. The orbitals are represented as p x, p y and p z. For example, for p-subshell, the value of m l will be, m l = – 1, 0, + 1. For a given value of l, the value of magnetic quantum number (m l) ranges form – l to + l. The magnetic quantum number (m l) represents the orbitals of a given subshell. Then in electron configuration it will be denoted as’2p 6‘. For example: If an atom is having 6 electrons with principal quantum number 2 in subshell of ‘p’. In electron configuration, the principal quantum number is stated just before the letter and the number of electrons with same orbital quantum number is represented as superscript of letter. For l = 2, shape of orbital is rounder ellipse with more value of angular momentum.įor different values of orbital or azimuth quantum number, the shape of orbitals is shown in table below. For l = 1, shape of orbital is an ellipse with some non-zero value of angular momentum. For Example: for orbital quantum number, l = 0, the value of angular momentum is zero and shape of orbital is straight line with zero angular momentum is. The orbital or azimuth quantum number represents the angular momentum and possible shape of orbital to which the electron is associated. Energy level or Orbit (shell) Principal quantum number ‘n’ Maximum Number of electrons (2n 2) 1 K 1 2×1 2=2 2 L 2 2×2 2=8 3 M 3 2×3 2=18 4 N 4 2×4 2=32 For different energy level (shells) the value of “Principal quantum Number ‘n’ and maximum number of electrons associate with different energy levels are given in table below- Sl. These energy levels are denoted by the letters K, L, M, N, ……. The electrons having principal quantum number, are associated with same energy levels (shells). Principal quantum number is used in Bohr and Summer field atomic model. Principal quantum number of an electron represents the main energy level or shell or orbit to which the electron belongs. Spin magnetic quantum number (m s) Principal Quantum Number (n).Orbital or Azimuthal quantum number (l).These quantum numbers are useful for representing the electron configuration. These quantum numbers represent the location, energy level and spin of electron in atom. /GettyImages-1157225833-f294a1f0fa314b12bf2da395e95107d7.jpg)
The quantum numbers are basically represents the address of an electrons in atom.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
